Helios EDC — Electronic Data Capture
Our proprietary clinical data capture ecosystem, validated per FDA 21 CFR Part 11 and GAMP 5 guidelines.
Request a Demo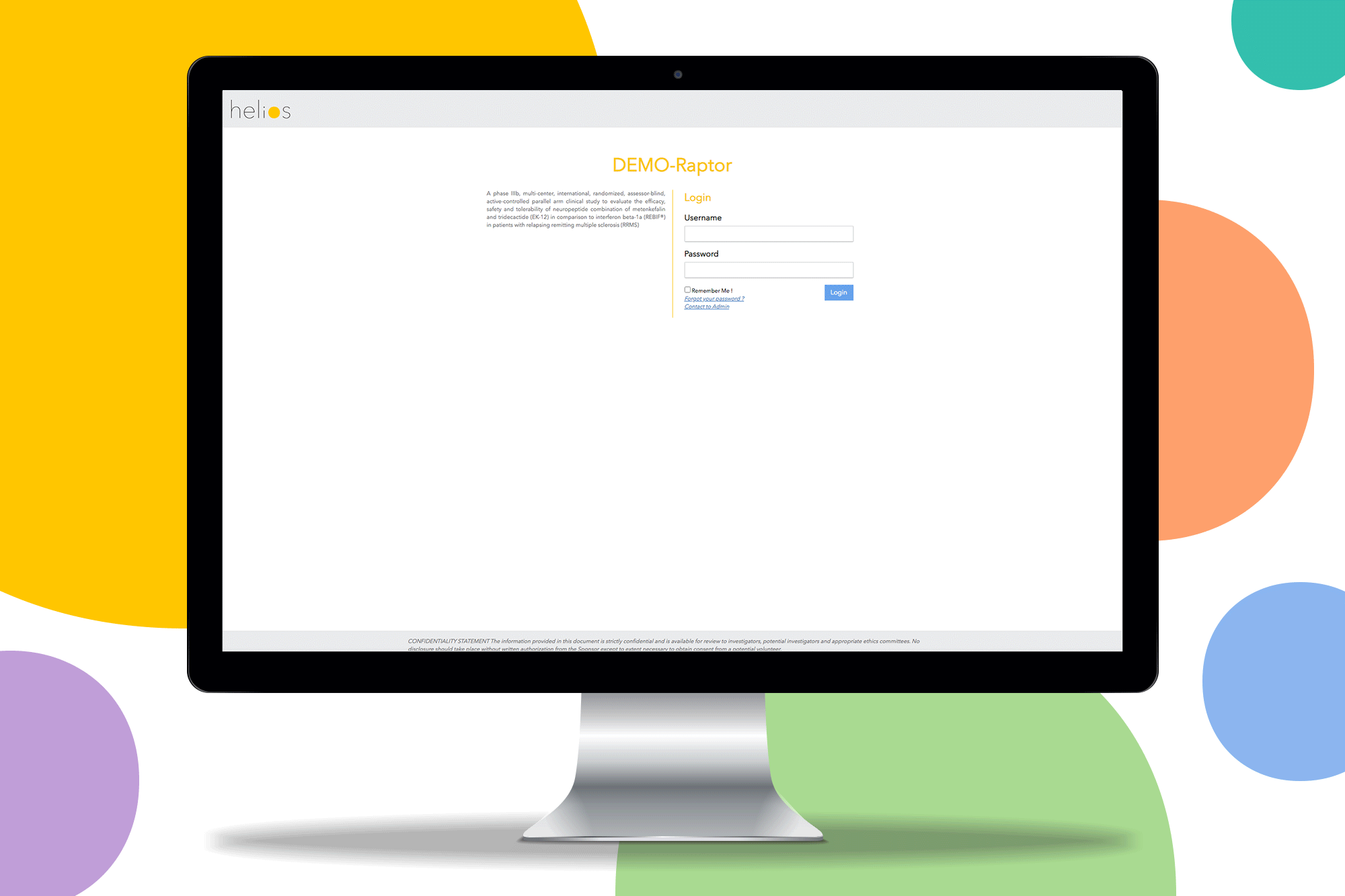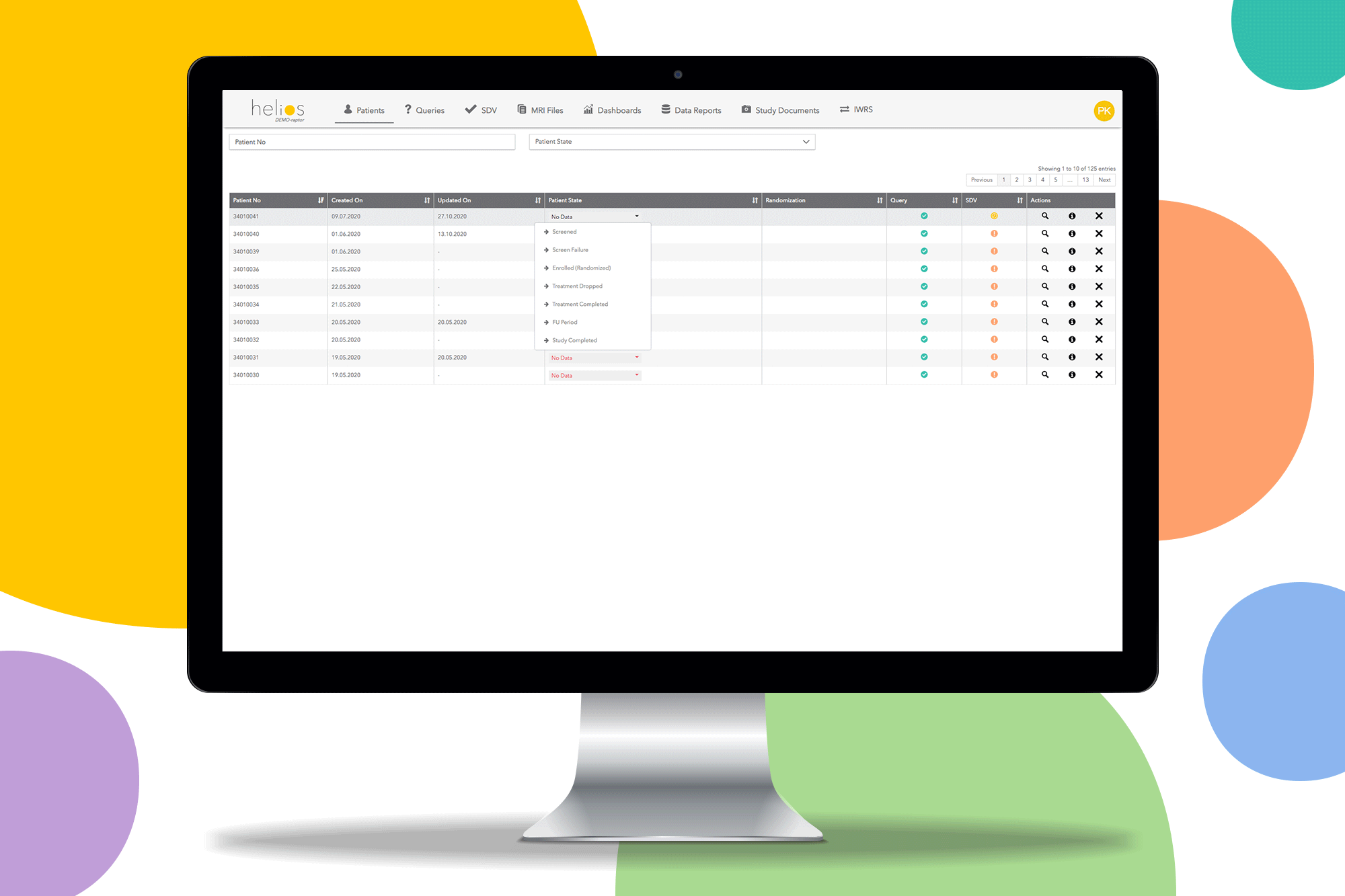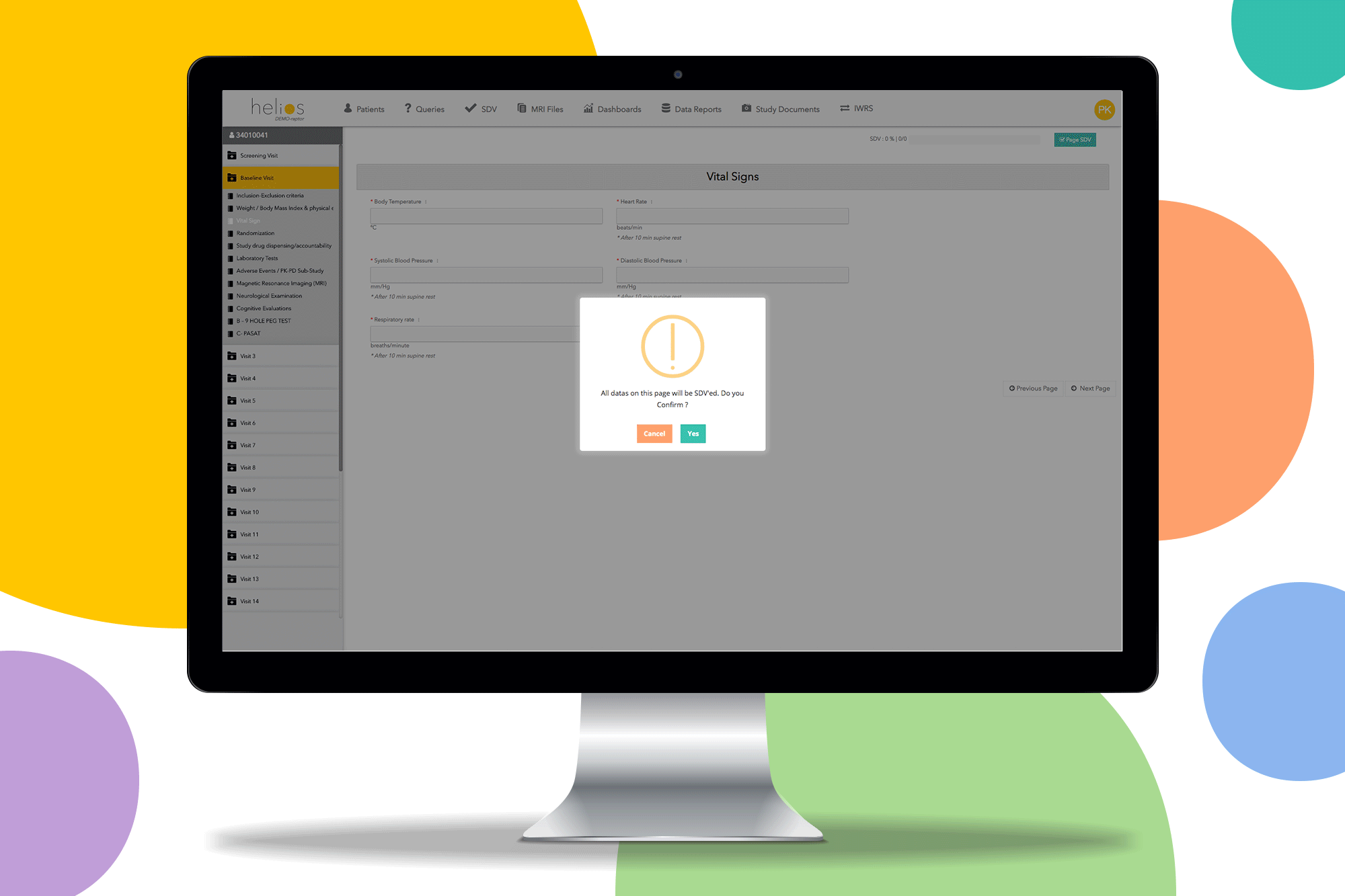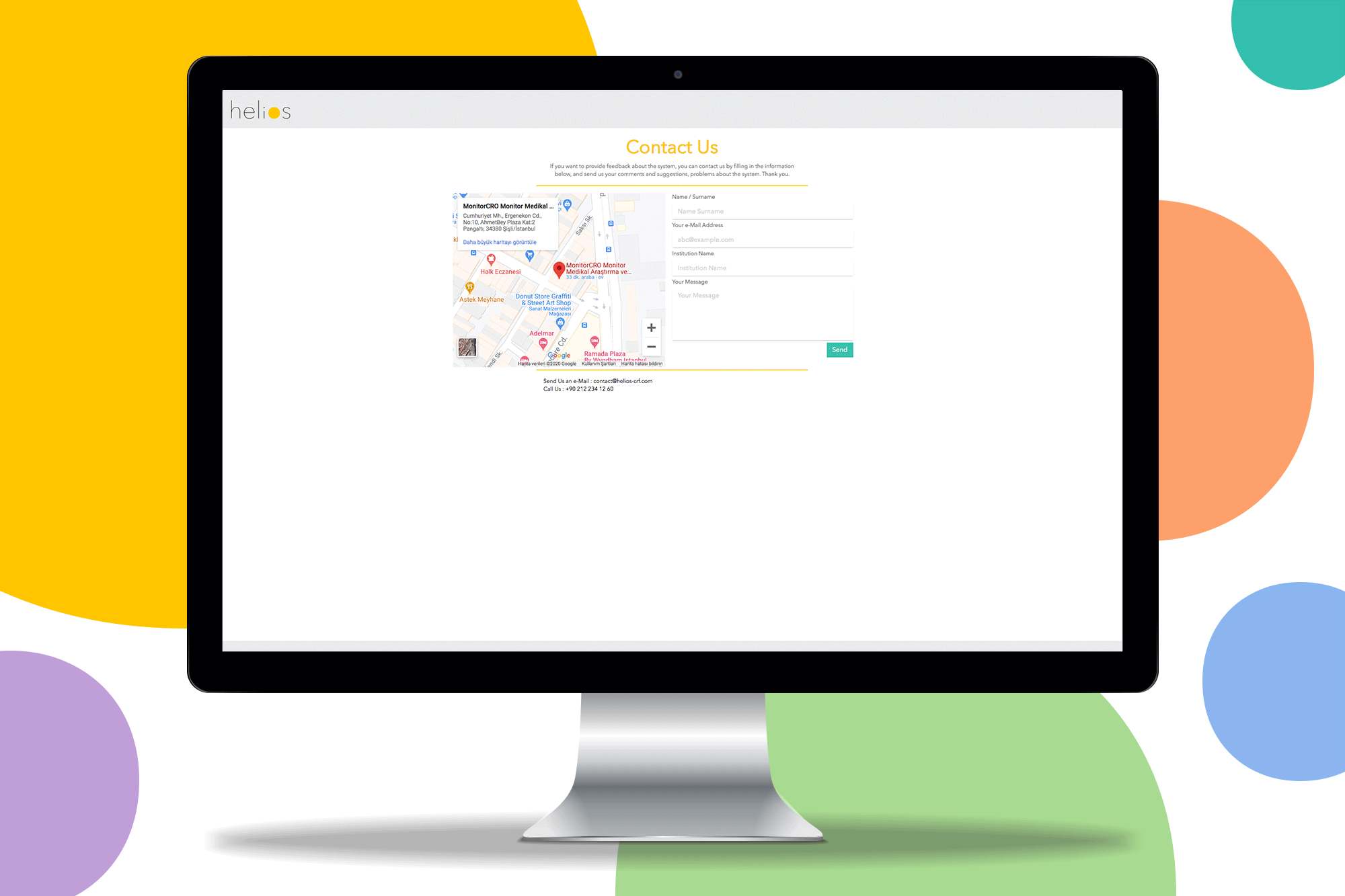
Platform Features
Built for Clinical Excellence
Study Designer
Drag-and-drop CRF builder with pre-built templates, allowing rapid study setup and flexible form design tailored to your protocol requirements.
Real-Time Data
Live dashboards and automated queries provide instant visibility into study progress, data quality metrics, and site performance.
User Access Management
Role-based access control with granular permissions ensures data security while enabling efficient collaboration across study teams.
Easy Monitoring
Source data verification (SDV) tools and remote monitoring capabilities streamline the monitoring process and reduce site burden.
Secure Validation Packages
Complete IQ/OQ/PQ validation packages with documented test scripts, ensuring full compliance with regulatory requirements.
Ultimate Flexibility
Configurable workflows, custom edit checks, and adaptable study designs support complex protocols across all therapeutic areas.
Randomization
Built-in randomization module supporting simple, block, and stratified randomization with double-blind capabilities and unblinding procedures.
Post-Market Surveillance (MDR)
Dedicated MDR module for medical device post-market surveillance, enabling continuous performance and safety data collection per EU MDR requirements.
Compliance & Validation
Computerized System Validation
Helios EDC is fully validated in accordance with FDA 21 CFR Part 11 and GAMP 5 guidelines. Our comprehensive validation approach ensures data integrity, security, and regulatory compliance throughout the clinical trial lifecycle.

GAMP 5 V-Model
Medical Devices
MDR Post-Market Surveillance
Helios EDC includes a dedicated module for Medical Device Regulation (MDR) post-market surveillance. Collect and manage post-market clinical follow-up (PMCF) data, track device performance, and generate periodic safety update reports (PSURs) — all within a single validated platform.
Request a Demo
See Helios EDC in action. Schedule a personalized demo with our team to explore how our platform can accelerate your clinical trials.
Request a Demo